What is Diabetes ?
The body is made of millions of cells that need energy to function. The food you eat is turned into sugar, or glucose. It is one of many substances needed by the cells to make energy. Glucose is carried to the cells through the bloodstream, with the help of insulin. Insulin is a chemical hormone, which is manufactured by the pancreas. It acts as a glucose transporter and provides glucose to the target cells of your body. Insulin levels in the blood vary with the amount of glucose present in the blood.
Diabetes is a chronic debilitating disease that makes it difficult for the cells of your body to get the glucose they need to make energy. As a result, the individual has high blood glucose (blood sugar), either because of inadequate production of insulin, or because of the unresponsiveness of the body's cells to insulin. Or in some cases, both.
If you have diabetes, your blood sugar level is altered. This altered blood glucose level can cause different problems with other bodily functions such as eyes, kidneys, nerves and feet functionality. Long term complications may include heart diseases, sexual dysfunction and bone and joint disorders.
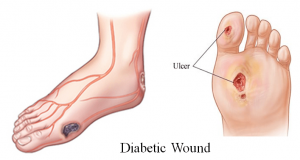
Diabetic Foot Wounds
Diabetic foot is one of the most dreadful complications of diabetes which can lead to prolonged hospitalization, restricted mobility, and sometimes limb amputation. The nerves become damaged due to poor blood circulation, resulting in delayed wound healing and high risk of infection. Burning pain, tingling sensations, weakness or loss of feeling are symptoms of diabetic foot. Because of sensation loss, temperature, pain and lack of attention to the feet become a danger. These become big risks from injury, unnoticed wounds and even ulcers. If left unnoticed, these wounds or ulcers can become infected, which is leads to serious conditions such as extended time in a care facility or limb amputations.
Diabetic patients with open wounds and infection should be treated aggressively due to the potential risks. The cornerstone of preventing these complications is to monitor the feet so that problems are spotted at the early stages. Other measures include keeping the feet clean and dry, ensuring the nails are well maintained and wearing socks and shoes that do not squeeze or rub the feet and fit comfortably.
Treatment of the diabetic wound comprises of five key factors which include wound assessment, tissue debridement, or removal of lifeless tissue from a wound, infection control, moisture balance and pressure reduction.
Diabetics are more prone to infections and therefore infections should be a top concern in any diabetic wound treatment plan. Different antibiotics and antiseptics are commonly used to reduce the growth of bacteria and their further spread. However, several recent studies have shown these methods might be causing more harm.
Topical antimicrobial or antiseptic agents like Povidone iodine and Hydrogen peroxide (H2O2) in chronic wounds remain in widespread use today and are successful in microbial eradication. However, they are found to be toxic to the cells involved in the wound healing process and their effectiveness varies between the types of microorganisms. Finding a non-cytotoxic, safe and effective wound cleanser was the biggest challenge for the physicians and healthcare staff. Hypochlorous acid was the solution.
Diabetic Wound Treatment with HOCL
Hypochlorous acid (HOCl) solution is one of the antiseptic agents used for the treatment of open diabetic wounds. It is a weak mixture of chemicals extensively used in deodorants and household bleach. It is naturally present in small molecules organically produced by white blood cells as a response to kill microorganisms before they invade the immune system. In recent years, clinical evidence shows HOCl to be safe with no negative effects on the cells of the body which take part in the wound healing process.
HOCl has a broad spectrum antimicrobial activity and is able to kill pathogens quickly. It is also effective in killing drug-resistant microorganisms. Studies have shown in addition to its antimicrobial activity, HOCl has the ability to remove strong odors from the wounds and reduces the discomfort of the wound by providing a cooling sensation.
Wound areas usually have a poor blood circulation, resulting in decreased oxygen supply, ultimately interfering with the healing process. HOCl increases the oxygenation of wound sites and supports the healing process. Another feature of HOCl is its significant anti-inflammatory activity in which it inhibits the degranulation of mast cells. This results in the reduction of pain and itching. Recent case studies have also shown the efficacy of HOCl in treating and managing the hypertrophic and keloid scars.
Ordinary HOCl anti-infective sprays contain a very small acid content and are suggested mainly to apply to a dressing. After wiping with HOCl and topping with a gentle rub of gauze, the healing time is improved with the wound losing unpleasant odors and becoming pain-free. Recommendations include the intralesional use of HOCl or other techniques that make sure the wound is covered with the solution for 15 minutes after debridement.
HOCl can also be used in the sterilization and debridement of the dead tissue of wounds observed at the source of injury.
Although the process of HOCl stabilization is very difficult, it is only a stabilized form of the acid that can help in cleansing wounds. A consultation with the physician is always necessary to see if it is a good idea to start using HOCl as a component of wound treatment.
In short, HOCl is a unique and powerful antimicrobial cleanser against a variety of pathogens. It is safe, painless, low cost and rapidly improves the wound healing, especially open wounds of a diabetic foot. HOCl also significantly reduces the use of systemic antibiotics and has an important role in keeping dermal matrices and skin grafts hydrated. Studies on the matter have recommended the use of HOCl as a reliable diabetic wound cleanser. More controlled clinical studies are required to evaluate the efficacy and safety of HOCl and the outcomes of various methods of its application.
"Hypochlorous Acid for Wound Care." Advanced Tissue. June 25, 2014. Accessed February 07, 2018. https://www.advancedtissue.com/hypochlorous-acid-wound-care/.
Biesman B, Cohen J, Gold M.
Integrating HOCl into Wound Care: A New Paradigm for Patient Management. Practical dermatology. PDF file. July, 2017.
www.practicaldermatology.com/pdfs/0717_insert.pdf.